Testing for thyroid cancer just got easier and less costly for millions of people across the country, thanks to technology developed by UPMC. The ThyroSeq® Genomic Classifier was recently approved for coverage by Medicare — the first step in making it available to more than 50 million people nationwide.
“We’ve spent the last 11 years optimizing this technology and making it better,” says lead investigator Yuri Nikiforov, MD, PhD, professor of pathology and director of UPMC’s Division of Molecular and Genomic Pathology. “Having it approved by Medicare is a big achievement. Patients can now have full access to the test.”
Never Miss a Beat!
Subscribe to Our HealthBeat Newsletter!
Thank you for subscribing!
You can now select the specific newsletters you'd like to receive.
You are already subscribed.
Subscribe to more newsletters in our email preference center.
Sorry, an error occurred. Please try again later.
Get Healthy Tips Sent to Your Phone!
Common Disease, Costly Testing
UPMC scientists developed the ThyroSeq test in the Molecular and Genomic Pathology Lab at UPMC. This next-generation, sequencing-based test evaluates thousands of genetic variants for cancer, or a malignancy, in the thyroid.
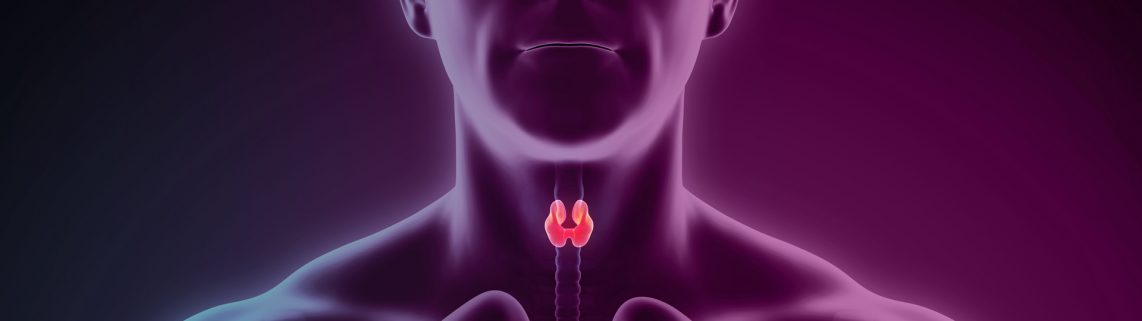
The thyroid is a small two-lobe gland in the neck that produces the hormones that regulate metabolism. It can develop nodules that may be cancerous, especially in women. The risk of thyroid cancer also increases with age, says Dr. Nikiforov. “After age 50, about half of all women have thyroid nodules,” he says. The American Cancer Society reports that nearly 54,000 people in the United States are diagnosed with thyroid cancer every year.
Of course, not all nodules are malignant. And thyroid cancer tends to develop slowly, so getting an early diagnosis can be a literal lifesaver. For years, the standard practice has been to insert a needle into the nodule to remove cells and then study them under a microscope to determine whether they’re cancerous. In 70 to 80 percent of cases, doctors can determine whether the cells are cancerous from that initial sample, says Dr. Nikiforov.
“But in the rest of the cases, the results are inconclusive. We cannot determine whether they are benign or malignant,” he notes. Before ThyroSeq, a patient with an ambiguous initial result had to undergo surgery — which involved removing at least half the thyroid — to determine if a nodule was cancerous.
“It was costly and patients often had to take synthetic hormones for the rest of their lives,” Dr. Nikiforov said. And in three out of four patients, the nodule proved to be benign.
Game-Changing Technology
ThyroSeq helps patients and doctors by reducing the need for unnecessary thyroid surgeries. Dr. Nikiforov explains that with ThyroSeq, a fine needle aspirator and a small tube are used to collect cells from the nodule, which are then stored in a freezer. “If the cytology is ambiguous, the patient doesn’t have to come back for another appointment. We can use the sample we’ve already collected,” says Dr. Nikiforov.
UPMC has offered ThyroSeq to its patients since 2014 and it has been used in thyroid clinics in all 50 states. Dr. Nikiforov says patients have come from as far as Singapore and Russia to access it.
The technology for ThyroSeq didn’t exist a decade ago. “We didn’t know all the genetic markers of thyroid cancer,” adds Dr. Nikiforov. “In the past decade, we’ve found thousands of gene variations.”
Dr. Nikiforov’s research continues as his team works to identify additional genetic markers for thyroid cancer: “We now have identified nearly 97 percent of all the genetic markers. We’re very close to that magic number of 100 percent.”
For more information about the services UPMC offers thyroid patients, visit the Multidisciplinary Thyroid Center or call 412-586-9205.
About UPMC Hillman Cancer Center
When you are facing cancer, you need the best care possible. UPMC Hillman Cancer Center provides world-class cancer care, from diagnosis to treatment, to help you in your cancer battle. We are the only comprehensive cancer center in our region, as designated by the National Cancer Institute. We have more than 70 locations throughout Pennsylvania, Ohio, and New York, with more than 200 oncologists – making it easier for you to find world-class care close to home. Our internationally renowned research team is striving to find new advances in prevention, detection, and treatment. Most of all, we are here for you. Our patient-first approach aims to provide you and your loved ones the care and support you need. To find a provider near you, visit our website.